
Hundreds of years earlier, Dalton had proposed that atoms were neutral, solid Positive plate of the tube and repelled by the negative plate of the same tube. Rays, cathode rays, which showed that they are composed of only negativelyĬharged particles which he called electrons, because they are attracted by the Of rays which travelled in straight lines from the cathode. Thomson was able to prove that the glow was due to some kind To 15000 volts, a bright green glow appeared on the glass. When the potential difference was increased 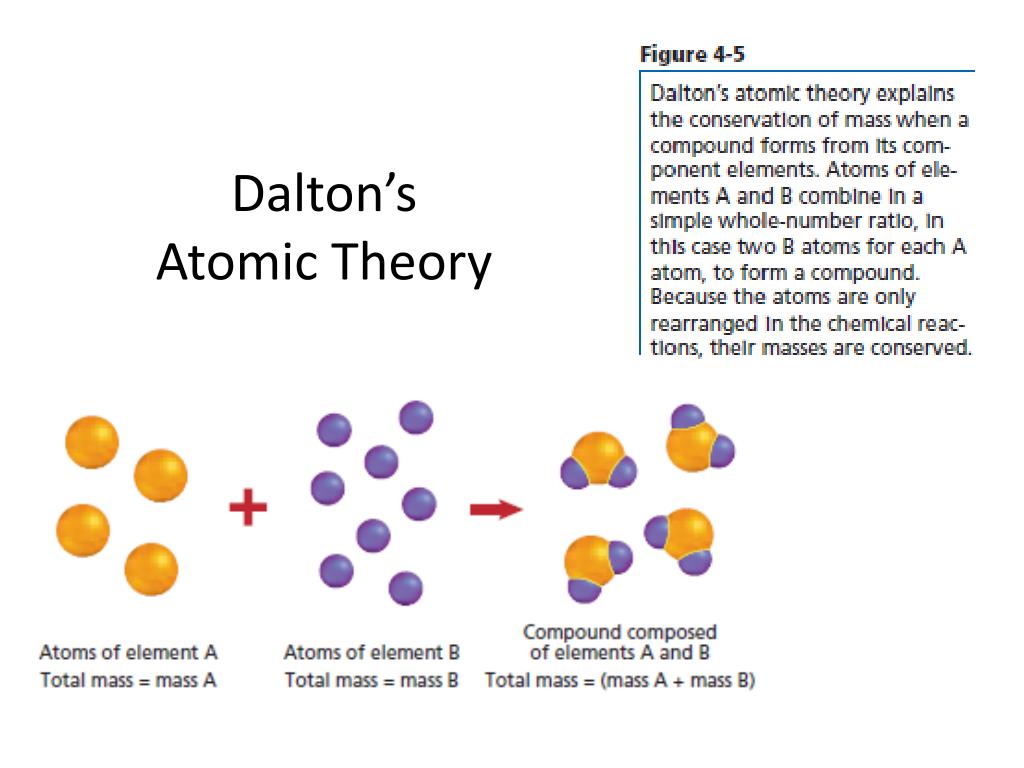
Tube containing a gas at a very low pressure of about 0.0001 atmospheric Showed that when a potential difference of 5000 volts was applied across a glass Using his research on Cathode Ray Tube technology, Thomson He helped to indirectly create the television and the computer. He was also the first scientist to show that the atom is made of other smaller particles and ended up contributing to more than the atomic theory. In 1897, as Cavendish Professor of Physics at Cambridge, Thomson discovered the electron which he had originally named as corpuscle. Thomson, physicist and inventor, 1897/ ThoughtCo. But in 1873, his father died and due to lack of funds changed his major to physics. Joseph John Thomson (1856–1940), the son of an English bookseller, had wanted to be an engineer and was already admitted to college at an unusually young age of 14 to study engineering. That were macroscopic, or seen with the unaided eye.Īlthough the two theories that proposed atoms couldn’t beĭivided were not true, John Dalton added significantly to the developments ofĪtomic theory, and would greatly influence J.J. His model was created solely on experiments That he came up with it without ever seeing the atom! He had no concept of The really awesome thing about Dalton’s model of the atom is 
Atoms of different elements are different. Atoms can neither be created nor destroyed.ģ. His atomic theory had five statements:īritish Chemist and Physicist, John Dalton (1766–1844)/ Biography.ġ. He was a schoolteacher who performed many experiments on atoms. The atom is now considered to be the basic unit of simple substances or elements. John Dalton (1766–1844), an English Chemist, put forward a theory to describe the nature of the atom. So, for the next two thousand years, the world depended on the theories of Democritus and Aristotle, until the year 1808, when John Dalton came up with the Modern Atomic Theory. Its actual existence was not established until the 19th century when the idea was accepted and refined by scientists. So, for the next 2,000 years, the world depended on the theories of Democritus and Aristotle, until the year 1808, when John Dalton came up with the Modern Atomic Theory.ĭue to a lack of evidence and experiments, their views on what atoms look like and how they behave were incorrect. His contribution helped people understand the idea of an atom and helped other scientists further look into the science of the atom and its generic makeup.ĭue to a lack of evidence and experiments, their views on what atoms look like and how they behave were incorrect. Democritus created the first atomic model (a round sphere with no electrons, protons, or neutrons). To them, human reasoning was superior to experimentation. 
Instead, they use reasoning to back up their beliefs. However, philosophers are not scientists nor do they test their ideas. These pursuits contributed to early chemical knowledge but were often shrouded in mysticism and superstition. Ancient Greeks considered Aristotle’s opinions as “very important” and if Aristotle thought the atomic idea had no merit, then most of these Greeks thought the same as well.ĭuring the Middle Ages and the Renaissance, scientists pursued various goals, including the transmutation of base metals into gold and the discovery of the philosopher’s stone. In fact, Aristotle dismissed the atomic idea as worthless. Unfortunately, the atomic ideas of Democritus had no lasting effects on other Greek philosophers, including Aristotle. Aristotle postulated that all matter was made of only four elements – Earth, Air, Water and Fire. Aristotle believed in the continuous and infinitely divisible nature of matter. He was the first person to use the word “atom”/ Life Persona.Īristotle, a prominent philosopher of the time, rejected the atomic theory in favour of his own ideas, which dominated scientific thought for centuries. Greek philosopher, Democritus (460–370 B.C.).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
